Exciting News: VYJUVEK is now approved for use from birth, can be applied by a caregiver or patient at-home, has an increased weekly volume, & has the flexibility of being removed at your next dressing change.
About VYJUVEK
Treating the genetic cause of DEB is important for achieving complete and long-lasting wound healing
VYJUVEK delivers working genes directly into skin cells, helping the body produce the missing protein needed to heal wounds.
VYJUVEK uses a carrier called a “vector” to deliver working genes directly into the skin cells.
Here's a simple way to picture how VYJUVEK works:

Think of a delivery car bringing skilled repair workers to a job site:
The car is the vector, and the workers it carries are genes with the tools needed to help heal wounds.
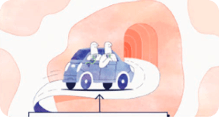
The car delivers the workers directly into the skin cells:
Once there, the workers get right to work, repairing the skin's structure from within.

But the job is never truly finished:
Because skin cells are always being replaced, new workers are needed, which is why you apply VYJUVEK until a wound closes and then re-apply as needed.
It's also important to note that VYJUVEK will not cause a herpes infection. VYJUVEK uses a herpes simplex virus (HSV) vector that has been changed so that it can't infect you. The safety of VYJUVEK was tested in a study, and no patients had active HSV infections.
The science behind how VYJUVEK works
Want more detail? Tap below to see how VYJUVEK delivers the missing genes that allow wounds to heal completely.
VYJUVEK delivers new COL7A1 genes directly to skin cells in DEB wounds
These COL7A1 genes restore the ability of those cells to make functional type VII collagen protein and form anchoring fibrils
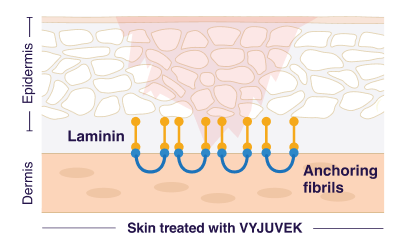
Anchoring fibrils bind the inner skin layer (dermis) and outer skin layer (epidermis) together and promote wound healing
In a clinical study, VYJUVEK was proven to heal and close wounds
The goal of the study was to evaluate complete wound healing (100% closure).
- The study included 31 people ranging in age from 1 to 44 years old with dominant DEB or recessive DEB
- For each person, 2 similar wounds were chosen. One was treated with VYJUVEK and the other received placebo
- Placebo is a treatment containing no medication that was given to participants the same way as VYJUVEK

- The main measure of the study was complete wound healing at 6 months
- Treatments were applied weekly until wounds were completely healed. Treatment resumed if wound reopened
After the 6-month study, 47 patients joined a long-term follow-up study for up to ~2.2 years that measured the safety and durability of healing.
VYJUVEK helped more people achieve complete wound healing
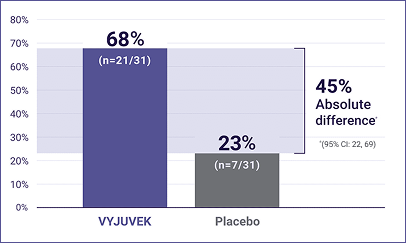
- At 3 months, 68% of wounds treated with VYJUVEK healed completely vs 23% of wounds treated with placebo
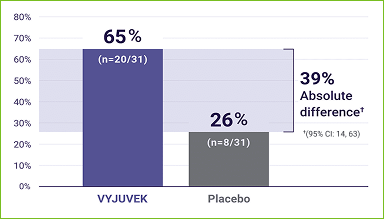
- At 6 months, 65% of wounds treated with VYJUVEK healed completely vs 26% of wounds treated with placebo
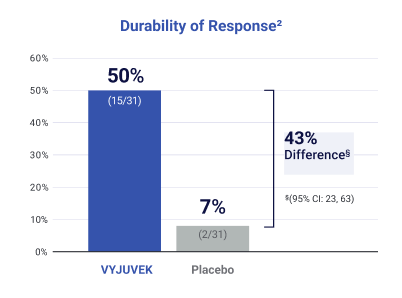
VYJUVEK helped wounds close and stay closed
VYJUVEK showed lasting results: 50% of treated wounds were fully healed at both 3 and 6 months, compared with only 7% of wounds treated with placebo.

“Another thing we’ve noticed is that as the area we’re treating is closing, the skin there has felt a lot stronger. Before I started treatment, if an area healed, it would usually be broken open again by the next week, because of how fragile that skin is. Since the treatment, I’d say around the second or third week, the skin has felt stronger.”
- Bianca, living with RDEB
VYJUVEK was well tolerated
| In the study of 31 people treated with VYJUVEK, these were the most common side effects: | |
|---|---|
| Side effects |
Number of people (%) |
| Itching | 3 (10) |
| Chills | 3 (10) |
| Redness | 2 (6) |
| Rash | 2 (6) |
| Cough | 2 (6) |
| Runny Nose | 2 (6) |

“In the beginning, I had
challenges with itching and feeling hot because I was using Saran™ Wrap, but then I changed my dressing to Mepitel, which helped stop those issues.”- Tristen, living with RDEB
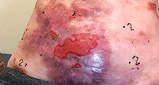
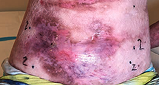
Across all wound sizes, VYJUVEK achieved complete wound closure
Stomach Wound
Living with RDEB 14 years old
Calf Wound
Living with DDEB 20 years old
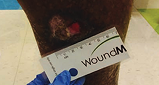
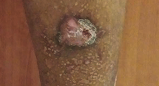
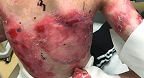
Back Wound
Living with RDEB 68 years old
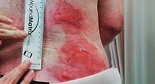
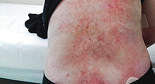
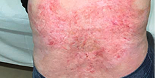

“The wounds that were treated with VYJUVEK seemed to heal fully, were less painful as well, and they stayed closed for a longer amount of time. There was a noticeable difference between the healing of those wounds versus the other non-treated ones.”
- Emily, living with RDEB
Over time, VYJUVEK has been shown to promote complete wound healing that lasts
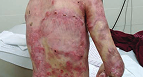
Thigh Wound
Living with RDEB 34 years old

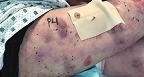
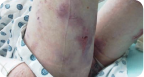
Knee Wound
Living with RDEB
8 years old
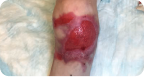
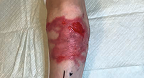
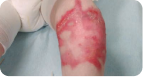
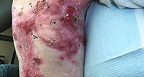
Back Wound
Living with RDEB 16 years old
VYJUVEK is a topical gel used to treat wounds in adult and pediatric patients (from birth) with dystrophic epidermolysis bullosa (DEB).
VYJUVEK gel should be applied by a healthcare professional, patient, or caregiver.
After treatment, patients and caregivers should be careful not to touch treated wounds and dressings until the next bandage change. If accidentally exposed to the VYJUVEK gel, clean the affected area.
Wash hands and wear protective gloves when changing wound dressings. Disinfect bandages from the first dressing change with a virucidal agent and dispose of the disinfected bandages in a separate sealed plastic bag in household waste. Dispose of the subsequent used dressings in a sealed plastic bag in household waste.
The most common adverse reactions (>5%) were itching, chills, redness, rash, cough, and runny nose. These are not all the possible side effects with VYJUVEK. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or to the Sponsor at 1-844-557-9782.
Please see Important Safety Information above and click here for full Prescribing Information.
VYJUVEK is a topical gel used to treat wounds in adult and pediatric patients (from birth) with dystrophic epidermolysis bullosa (DEB).
VYJUVEK gel should be applied by a healthcare professional, patient, or caregiver.
After treatment, patients and caregivers should be careful not to touch treated wounds and dressings until the next bandage change. If accidentally exposed to the VYJUVEK gel, clean the affected area.
Wash hands and wear protective gloves when changing wound dressings. Disinfect bandages from the first dressing change with a virucidal agent and dispose of the disinfected bandages in a separate sealed plastic bag in household waste. Dispose of the subsequent used dressings in a sealed plastic bag in household waste.
The most common adverse reactions (>5%) were itching, chills, redness, rash, cough, and runny nose. These are not all the possible side effects with VYJUVEK. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or to the Sponsor at 1-844-557-9782.
Please see Important Safety Information above and click here for full Prescribing Information.

